요약고중성지방혈증에 의한 임신성 췌장염의 대부분은 고지혈증 환자가 임신으로 인하여 과체중이 되거나 중성지방과 관련된 이차적인 인자를 가질 때 발생한다. 지금까지 국내에서 고중성 지방혈증에 의한 임신성 췌장염은 6예가 보고되었으나 그중 중성지방 수치가 1,000 mg/dL 이하인 적은 없었다. 임신 30주된 36세 여성이 상복부 통증을 주소로 입원하였다. 말초혈액 검사에서 중성지방 수치는 260 mg/dL, 아밀라아제 수치는 2,951 U/L, 리파아제 수치는 3,500 U/L였다. 복부 초음파 검사에서 저에코성 rim을 동반한 췌장의 부종이 관찰되었다. 며칠 후, 환자는 퇴원하여 임신 38주에 정상 분만을 하였다. 지단백질 전기 영동 결과는 IV형 고지단백혈증과 일치하였다. 이 증례보고는 이전에 보고된 고중성 지방혈증에 의한 임신성 췌장염과 달리, 중성지방 수치가 500 mg/dL 미만이지만, IV형 고지단백혈증의 존재시 발생한 급성 췌장염을 처음으로 보고한 것이다.
AbstractMost cases of hypertriglyceridemia (HTG)-induced gestational pancreatitis occur when a person with hyperlipidemia is overweight due to pregnancy or has secondary triggers associated with triglycerides (TGs). In Korea, 6 cases of HTG-induced gestational pancreatitis have been reported, but none of the affected patients had TG levels below 1,000 mg/dL. A 36-year-old female at 30 weeks of gestation was admitted due to pain in her upper abdomen. Initial biochemical analysis revealed a TG level of 260 mg/dL, an amylase level of 2,951 U/L and a lipase level of 3,500 U/L. Abdominal ultrasonography showed pancreatic swelling with a hypoechogenic rim. After several days, the patient was discharged and had a normal delivery at 38 weeks of gestation. This case report is the first to describe acute pancreatitis occurring in the presence of type IV hyperlipoproteinemia even though the TG level was less than 500 mg/dL, contrary to findings in previously reported cases.
INTRODUCTIONPancreatitis is a common cause of abdominal pain [1], but the incidence of pancreatitis during pregnancy is very low [2]. However, pancreatitis caused by pregnancy may cause premature labor and fetal death. Hypertriglyceridemia (HTG)-induced pancreatitis has been reported in 1 to 14% of acute pancreatitis cases and in up to 56% of acute pancreatitis cases occurring during pregnancy [3-5]. Pancreatitis can occur due to increased estrogen at the time of pregnancy, but the incidence is very low; even when pancreatic occurs, obstetricians often do not recognize it. Most cases of HTG-induced gestational pancreatitis occur when a person with hyperlipidemia is overweight due to pregnancy or has secondary triggers associated with triglycerides (TGs) [6]. In Korea, six cases of HTG-induced gestational pancreatitis have been reported, but none of the affected patients had (TG) levels below 1,000 mg/dL [6]. We report a case of acute pancreatitis diagnosed as acute pancreatitis with type IV Hyperlipoproteinemia during the third trimester of pregnancy and review relevant literature.
CASEA 36-year-old female visited the emergency room of Daejeon Eulji University Hospital for upper abdominal pain. The patient was at 30 weeks of gestation and gravida 2, para 2. During her previous two pregnancies, she experienced abdominal pain at 8 months of gestation similar to her pain at hospital admission, but she improved after several days of observation. This time, she visited another obstetrical clinic 1 day after symptom onset and her amylase levels were increased; thus, she visited the emergency room. Her mother was taking medication for hyperlipidemia.
Physical examination at the time of admission revealed an acutely ill appearance. The patient’s height was 158 cm, and her weight was 47 kg. Her vital signs were a blood pressure of 115/64 mmHg, a heart rate of 95 beats/min, a respiratory rate of 20 breaths/min and a body temperature of 36.3℃. The patient complained of pain and tenderness in the upper abdomen, and her pulmonary, cardiac, and bowel sounds were nonspecific. Initial biochemical analysis revealed a white blood cell count of 11,720/mm3, a hemoglobin level of 12.0 g/dL, a platelet count of 239 × 103/L, an aspartate transaminase level of 18 IU/L, an alanine transaminase level of 14 IU/L, an alkaline phosphatase level of 102 IU/L, a gamma-glutamyl transpeptidase level of 6 IU/L, a blood urea nitrogen level of 4 mg/dL, a calcium level of 9.7 mg/dL, a blood glucose of 142 mg/dL, a hemoglobin A1C of 4.9%, a total cholesterol level of 262 mg/dL, a high-density lipoprotein cholesterol level of 94 mg/dL, a low-density lipoprotein cholesterol level of 116 mg/dL, a TG level of 260 mg/dL, a C-reactive protein level of 2.34 mg/dL, an amylase level of 2,951 U/L and a lipase level of 3,500 U/L (Table 1). Her lipid profile was tested after admission.
Abdominal ultrasonography was performed to identify the etiology of the abdominal pain. No abnormal findings were observed in the liver, gallbladder or bile duct. Pancreatic swelling with a hypoechoic rim was observed, suggesting acute pancreatitis (Fig. 1). After admission, fasting and fluid therapy were initiated, and the Bedside index for severity in acute pancreatitis (BISAP) score was calculated within 24 hours. The patient’s BISAP score was 0 points, and mild pancreatitis was suspected. We observed no contractions of the uterus on obstetrical examination. After 5 days, the patient’s abdominal pain improved, and she could begin to eat again.
After starting to eat again, the patient had no abdominal pain, and her amylase and lipase levels improved to 88 U/L and 33 IU/L, respectively. She was discharged on the 8th day after admission. The patient had a natural delivery at 38 weeks of gestation two months after discharge. One month after spontaneous labor, a blood test was performed at the obstetrical clinic, and the patient visited our hospital again and had a TG level of 404 mg/dL. Lipoprotein electrophoresis was performed due to the patient’s family history. Apolipoprotein B measured 148 mg/dL (60–117 mg/dL), and apolipoprotein C-3 measured 21.9 mg/dL (5.5–9.5 mg/dL). The lipoprotein electrophoresis results were consistent with type IV hyperlipoproteinemia (Fig. 2). The patient did not breastfeed and was prescribed a combination of atorvastatin and ezetimibe orally, followed by outpatient monitoring.
DISCUSSIONPancreatitis in pregnancy is rare, with an incidence of 0.03% [2]. Acute pancreatitis during pregnancy is usually accompanied by secondary causes, such as gallstones [2]. HTGinduced pancreatitis has been reported in 1 to 14% of acute pancreatitis cases and in up to 56% of acute pancreatitis cases during pregnancy [3-5]. Elevated cholesterol levels are not associated with pancreatitis [7]. Serum TGs peaked at the 3rd trimester during pregnancy; TG levels are rarely found to be greater than 300 mg/dL (3.3 mmol/L). If serum triglyceride levels are >500 mg/dL (5.6 mmol/L), the risk of acute pancreatitis begins to rise and is markedly increased at levels above 1,000 mg/dL (11.3 mmol/L). The incidence of acute pancreatitis has been reported to be approximately 5% for TG levels >1,000 mg/dL (11.3 mmol/L) and 10–20% for TG levels >2,000 mg/dL (22.6 mmol/L) [8-11]. Studies involving patients with familial HTG with long-term follow-ups have shown that in the absence of other causes of pancreatitis, extreme elevations in TG levels occur during episodes of acute pancreatitis and are known as a so-called “hyperlipidemic abdominal crisis.” [12] A TG level higher than 1,000 mg/dL is generally believed to precipitate episodes of acute pancreatitis [7].
Two types of HTG exist: genetic and acquired. The most frequent cause of HTG is an abnormality in regulation of endogenous production of TG-rich very low-density lipoprotein (VLDL). This abnormality can cause either elevated VLDL alone (type IV hyperlipoproteinemia) or elevated VLDL and chylomicrons (type V hyperlipoproteinemia). Endogenous HTG is usually related to hyperinsulinemia and insulin resistance and is most often caused by obesity, excess calorie intake, alcohol use, or the use of estrogens or certain medications [13]. Type IV Hyperlipoproteinemia is an autosomal dominant disorder. This relatively common phenotype is characterized by high TG levels due to an isolated elevation of VLDL particles, which results from both overproduction and decreased elimination of these particles [10,14,15]. Type IV Hyperlipoproteinemia associated with moderate elevations in the serum triglyceride concentration (300 to 900 mg/dL) [16]. Types I and V hyperlipoproteinemia can present with spontaneous pancreatitis in the absence of a secondary factor; however, type IV hyperlipoproteinemia almost always requires a secondary factor to increase TG levels substantially [13]. Estrogen may promote hyperlipidemia by promoting VLDL production in the liver, and type IV HTG-induced pancreatitis may be caused by estrogen elevation due to pregnancy [14].
According to a previous study, hyperlipidemic pancreatitis was associated with especially poor outcomes compared to pancreatitis of other etiologies and with significantly increased rates of preterm delivery and pseudoocysts [2]. In Korea, six cases of gestational pancreatitis due to HTG have been reported. The serum TG levels of these six patients were higher than 1,000 mg/dL, and only one patient had a full-term birth (Table 2) [6].
In this case, the patient did not suffer from pancreatitis before pregnancy. Although she did not have blood tests or imaging performed during her first or second pregnancy, she complained of similar symptoms during the latter stage of each pregnancy. During her third pregnancy, she had a similar complaint of abdominal pain, and we were able to diagnose pancreatitis according to her obstetrician’s suspicion. After discharge, abdominal computed tomography showed no abnormalities of the pancreatic duct or structural abnormalities. We did not perform an autoimmune test. At the time of admission, endoscopic ultrasonography (EUS) was not readily available in our center. After discharge, the patient refused EUS and magnetic resonance cholangiopancreatography because of breastfeeding, reflecting a limitation due to insufficient testing for the cause of pancreatitis.
The patient’s TG levels were lower than those reported in previous cases of gestational HTG-induced acute pancreatitis [6]. However, ultrasonography revealed no evidence of gallstones, and type IV hyperlipoproteinemia was confirmed by lipoprotein electrophoresis. This case report is the first to describe acute pancreatitis occurring in the presence of type IV hyperlipoproteinemia even though the TG level was less than 500 mg/dL, contrary to findings in previously reported cases.
No reports indicate that TG levels are related to prognosis in terms of delivery, but the risk of acute pancreatitis has been reported to be increased in association with elevated TG levels [17]. This patient’s TG levels were lower than those reported in other patients. The patient was discharged with supportive care and delivered naturally at 38 weeks of gestation, suggesting that TG levels are related to patient prognosis. Future large-scale studies are required to verify this relationship.
Our study has certain limitations. We were unable to perform EUS at the time of admission and after discharge due to patient refusal. Ultrasonography cannot completely evaluate gallbladder microlithiasis, the distal common bile duct, or anatomical abnormalities such as pancreatic divisum. Evaluation for autoimmune pancreatitis is also necessary.
If a patient experiences persistent pain during pregnancy and no specific findings are observed on routine laboratory tests or uterine ultrasound, the possibility of pancreatitis should be considered. In gestational pancreatitis, because there is a limitation of the imaging, the examination for the differential diagnosis such as TG level should be done. In this patient, pancreatitis may have been present during her first and second pregnancies. If pregnant women complain of refractory abdominal pain, laboratory testing should be considered, including measurements of amylase and lipase levels and abdominal ultrasound.
REFERENCES1. Liljekvist MS, Pommergaard HC, Burchar th J, Rosenberg J. Diagnostic imaging and acute abdominal pain. Ugeskr Laeger 2015;177:V06140339.
2. Eddy JJ, Gideonsen MD, Song JY, Grobman WA, O’Halloran P. Pancreatitis in pregnancy. Obstet Gynecol 2008;112:1075-1081.
3. Chang CC, Hsieh YY, Tsai HD, Yang TC, Yeh LS, Hsu TY. Acute pancreatitis in pregnancy. Zhonghua Yi Xue Za Zhi (Taipei) 1998;61:85-92.
4. Fortson MR, Freedman SN, Webster PD 3rd. Clinical assessment of hyperlipidemic pancreatitis. Am J Gastroenterol 1995;90:2134-2139.
5. Scherer J, Singh VP, Pitchumoni CS, Yadav D. Issues in hypertriglyceridemic pancreatitis: an update. J Clin Gastroenterol 2014;48:195-203.
6. Park JM. Hypertriglyceridemia-induced acute pancreatitis. Korean J Pancreas Biliary Tract 2017;22:158-164.
7. Toskes PP. Hyperlipidemic pancreatitis. Gastroenterol Clin North Am 1990;19:783-791.
8. Athyros VG, Giouleme OI, Nikolaidis NL, et al. Long-term follow-up of patients with acute hypertriglyceridemia-induced pancreatitis. J Clin Gastroenterol 2002;34:472-475.
9. Basar R, Uzum AK, Canbaz B, et al. Therapeutic apheresis for severe hypertriglyceridemia in pregnancy. Arch Gynecol Obstet 2013;287:839-843.
10. Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive Summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA 2001;285:2486-2497.
11. Feldman M, Fried man LS, Sleisenger MH. Sleisenger and Fordtran’s gastrointestinal and liver disease: pathophysiology, diagnosis, management. 7th ed. Philadelphia, Saunders, 2002.
12. Miller A, Lees RS, McCluskey MA, Warshaw AL. The natural history and surgical significance of hyperlipemic abdominal crisis. Ann Surg 1979;190:401-408.
13. Yadav D, Pitchumoni CS. Issues in hyperlipidemic pancreatitis. J Clin Gastroenterol 2003;36:54-62.
14. Berglund L, Brunzell JD, Goldberg AC, et al. Evaluation and treatment of hypertriglyceridemia: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 2012;97:2969-2989.
15. Johansen CT, Hegele RA. Allelic and phenotypic spectrum of plasma triglycerides. Biochim Biophys Acta 2012;1821:833-842.
16. Yuan G, Al-Shali KZ, Hegele RA. Hypertriglyceridemia: its etiology, effects and treatment. CMAJ 2007;176:1113-1120.
17. Sandhu S, Al-Sarraf A, Taraboanta C, Frohlich J, Francis GA. Incidence of pancreatitis, secondary causes, and treatment of patients referred to a specialty lipid clinic with severe hypertriglyceridemia: a retrospective cohort study. Lipids Health Dis 2011;10:157.
18. Choi HS, Park JH, Kim JY, Im MW, Lee BI, Lee WY. Hypertriglyceridemia induced acute pancreatitis in pregnancy. Korean J Obstet Gynecol 2008;51:1514-1518.
19. Kang JY, Choi GY, Jeong HS, et al. Hypertriglyceridemia-induced acute pancreatitis in pregnancy: a case report. Korean J Med 2009;77:104-108.
20. Kim GH, Jun HA, Song JE, Lee KY, Kim SS. A case of acute pancreatitis induced by hypertriglyceridemia in gestational diabetes. Korean J Obstet Gynecol 2010;53:535-539.
21. Lee YJ, Park DS, Kim TH, Cho JH. A case of hypertriglyceridemia induced gestational pancreatitis treated by plasma exchange. Korean J Blood Transfus 2011;22:70-75.
Fig. 1.Ultrasonographic findings of gallbladder and pancreas. (A) No specific finding such as gallstones was noted. (B) Diffuse swelling (arrows) of the pancreas with a hypoechoic peripancreatic rim (arrowheads). 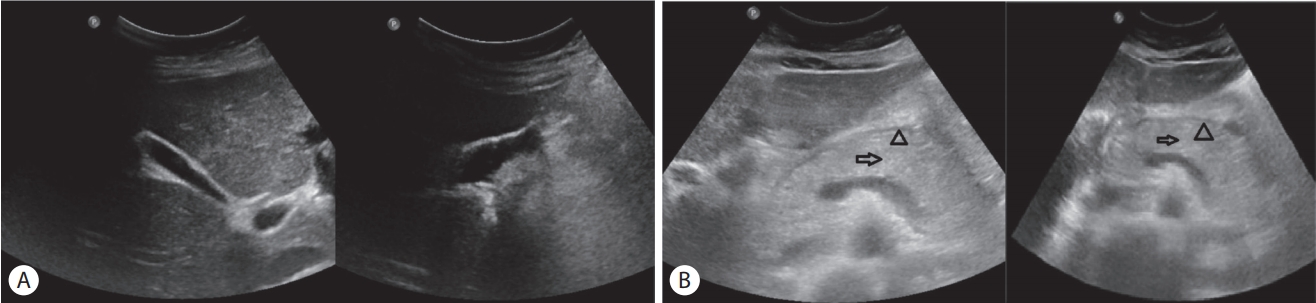
Fig. 2.Lipoprotein electrophoresis showing increased pre - beta and chylomicron fractions consistent with t ype IV hyperlipoproteinemia. Ref., reference. 
Table 1.The patient’s laboratory findings Table 2.HTG-induced pancreatitis in pregnancy cases in Korea
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||