INTRODUCTION
Cat-scratch disease (CSD) is a zoonotic infection caused by the bacterium Bartonella henselae that typically affects children and young adults. The most common manifestations of CSD include an erythematous papule at the site of cat scratch or bite, lymphadenopathy proximal to the scratch or bite location, and fever, which accounts for approximately 90% of all cases. However, atypical manifestations of this disease can affect various parts of the body and may result in more severe illness. Liver and splenic lesions due to CSD are considered rare, occurring in less than 10% of cases. In this case report, we present a patient with atypical manifestations of CSD causing liver tumor. This case was collected retrospectively and already been long enough, so the informed conset could not be obtained.
CASE
A 57-year-old woman experienced intermittent pain in the right upper abdomen and epigastrium for 3 months and got worse day by day. Additionally, the patient had lost 11 kg, and over the past few weeks, she frequently complained of bloating and nausea. She had no previous history of hepatitis or malignancies and never had a cat.
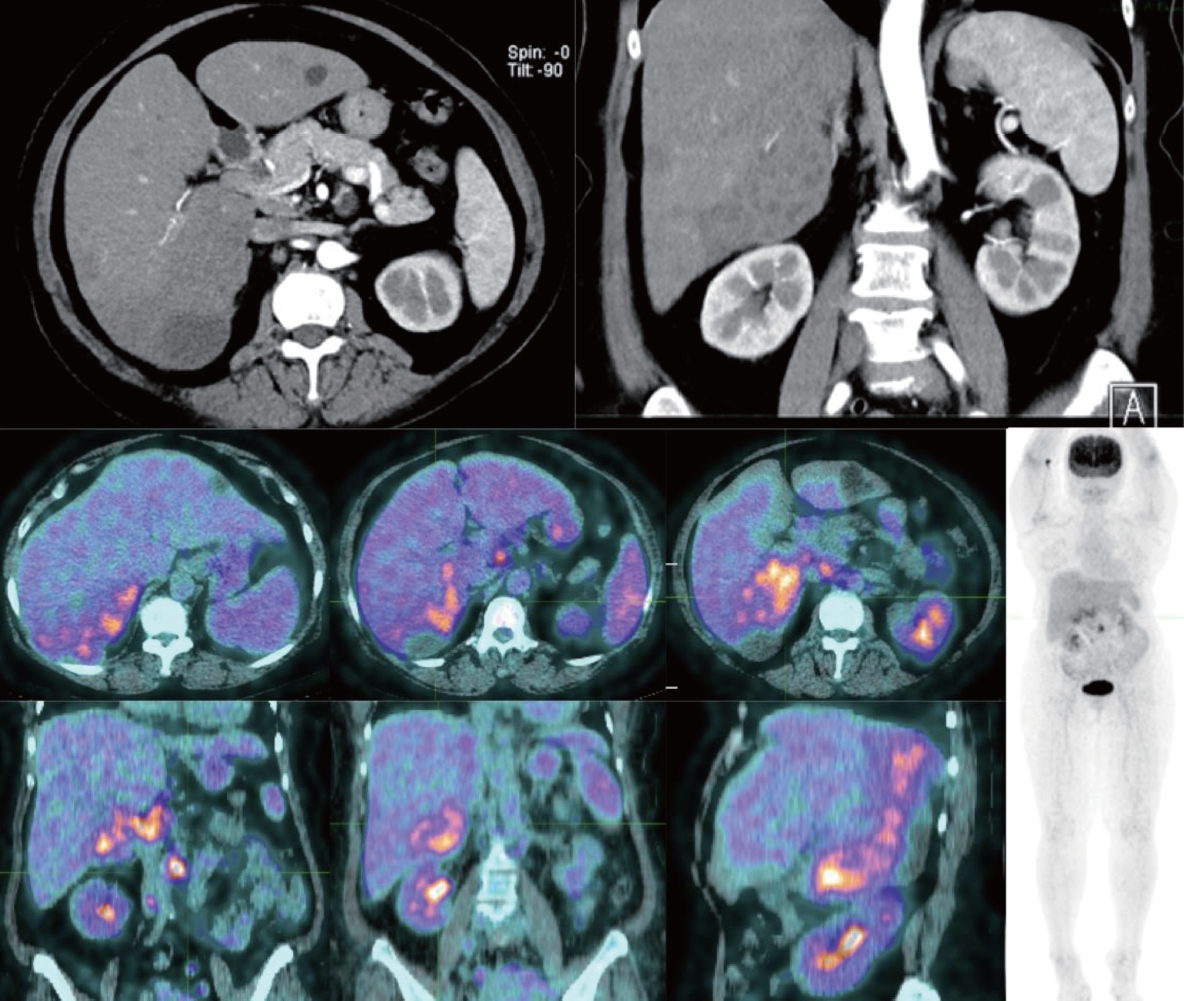
When she felt the pain for the first time, she was treated at another hospital and an abdominal ultrasound was performed. They found heterogeneous lesions in the right lobe of the liver and cysts in the left lobe of the liver. The patient was treated conservatively with medications but showed no change in subsequent follow-up ultrasound. Then she came to our hospital bringing her fluorodeoxyglucose (FDG) positron emission tomography (PET)/computed tomography (CT) scan results, which showed right liver lobe mass with inhomogenous FDG uptake and tissue density (8.66×9.76 cm) highly suspicious for malignancy, multiple FDG avid lymph nodes in the porta hepatica, pancreaticohepatic, paraaortic, and aortocaval suggesting metastases, and multiple hypodense non FDG avid nodules in left liver lobe consistent with benign lesions (cysts) (Fig. 1).
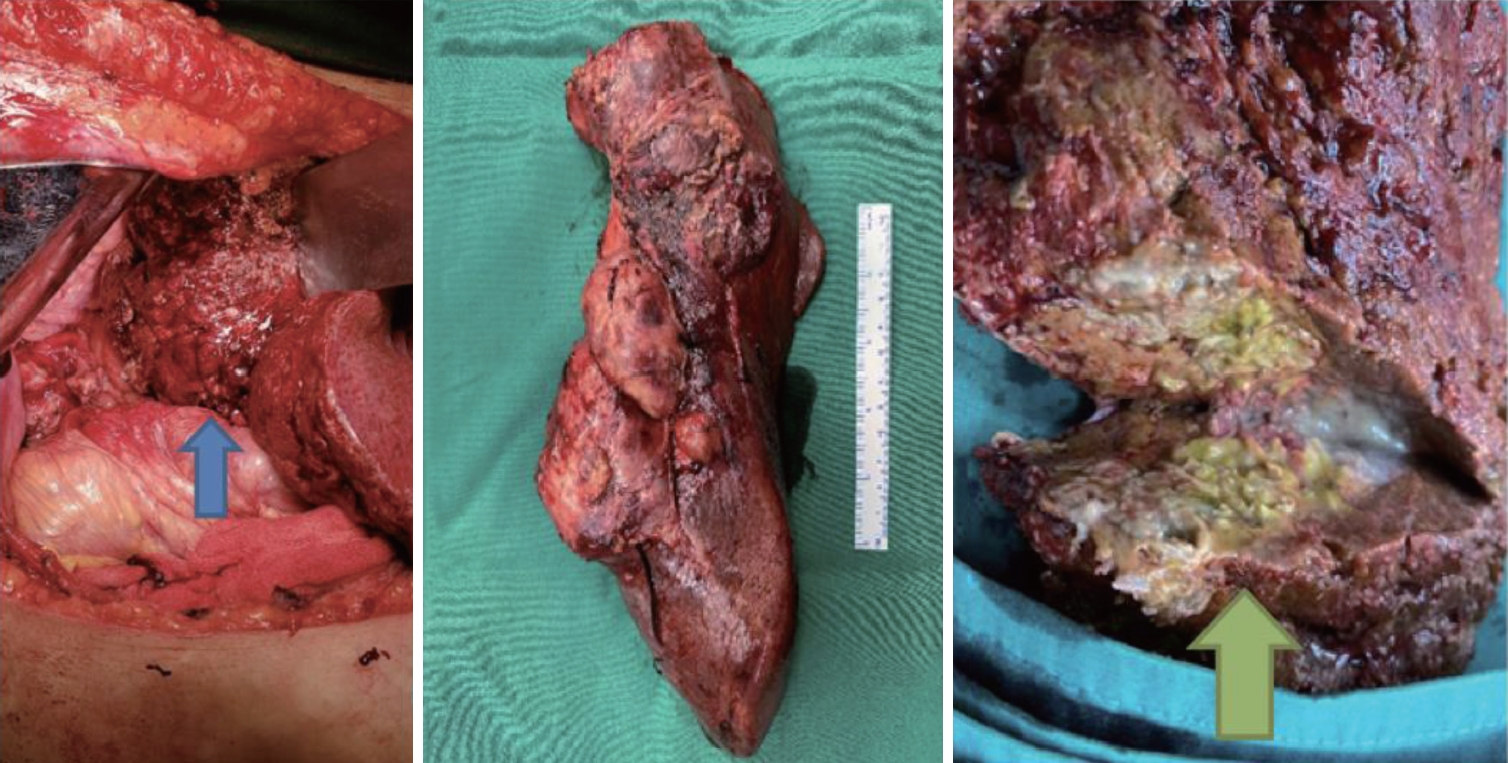
Although the liver function, carcinoembryonic antigen, and alpha-fetoprotein were all within normal limits, as the FDG PET/CT scan implies, we still could not rule out intrahepatic cholangiocarcinoma (ICC) as the primary diagnosis, and thus the patient was scheduled for liver resection. During the surgery, we found abnormal liver tissues from segments VI-VII measuring 26×12×10 cm with a chewy and solid consistency and some areas appeared to be bumpy (Fig. 2). When we dissected it, the tumor mass appeared gray, yellow, and greenish, and some nodules were noticed. The specimen was then sent for histopathological analysis along with three other tissues: hepatoduodenal lymphadenopathy, cysts from the liver’s left lobe, and the gallbladder. One gram of meropenem was administered intravenously every 8 hours postoperatively. The patient’s condition was stable following surgery, and she was discharged five days later with oral cefixime 200 mg twice daily for one week.
The patient’s condition was good during all of her postoperative visit, with surgical pain under control and there was no other major complaints. Histopathological examination showed necrotizing granulomatous hepatitis/CSD, reactive lymphoid hyperplasia of hepatoduodenal lymph nodes, biliary cysts, and active chronic cholecystitis, with no evidence of malignancy and mycobacterial infection (Fig. 3). The patient confirmed that she does not have a cat, but she has played with her neighbour’s cat on several occasions.
DISCUSSION
CSD is the most common human infection caused by the bacterium B. henselae [1]. Cats are the primary reservoir for B. henselae , and the Gram-negative bacterium is transmitted to humans by scratches or bites of infected cats [2].
Children and adolescents are more frequently affected by CSD [3]. Epidemiological studies suggest that children <14 years of age account for 32.5% of all CSD cases with the highest incidence found in children 5-9 years of age (9.4 cases/100,000) [2]. Among adults, the highest incidence was found among women aged 60-64 years (6.6 cases/100,000 for outpatients and 0.3 cases/100,000 for inpatients) [4].
In immunocompetent patients, subacute regional lymphadenopathy and fever are common findings, which were absent in our patient [3,5]. A surveillance study in Israel demonstrated that elderly patients had a lower incidence of lymphadenopathy and were more likely to present with general malaise and other atypical manifestations, including hepatosplenomegaly [6]. Unusual forms of CSD involving ocular, neurological, musculoskeletal, and hepatosplenic manifestations are rarely documented, ranging from 0.3 to 10% of all CSD cases [7]. Hepatosplenic lesions in CSD are considered very uncommon, affecting 0.3-0.7% of patients, and are more often described in children [6].
Both typical and atypical manifestations of CSD are associated with laboratory abnormalities including leukocytosis and elevated levels of inflammatory markers such as erythrocyte sedimentation rate and C-reactive protein [2,5]. Hepatosplenic involvement may be associated with normal or mildly increased transaminases [3,5]. In our case, the patient presented with normal transaminase and other liver function levels.
CSD diagnosis was originally made on the basis of clinical criteria such as (1) a history of cat contact, cat scratch, or other inoculation events; (2) positive cat scratch skin test reaction; (3) local lymphadenopathy with no other apparent etiology; and (4) typical histopathologic feature on biopsy [8]. However, atypical manifestations may require additional examination. Hepatic manifestations of CSD can be further inspected in imaging studies as non-specific hypoechoic liver nodules on ultrasonography, or persistent hypoattenuation relative to the liver and rim enhancement on contrast-enhanced CT scan [9,10]. Ultrasonography has a sensitivity of 83-87% in detecting hepatic lesions, compared to the 94-100% sensitivity of CT scan [7].
Clinical manifestations, laboratory work-up, and radiology examinations in our patient were inconclusive. The patient was relatively healthy with no remarkable risk factors. However, she came with a chronic condition going on for months, along with moderate weight loss. Our suspicion of malignancy was strongly supported by her readily-available abdominal ultrasound and PET/CT scan result showing the presence of liver mass and multiple lymph nodes with high FDG uptake. It is well known that cell with enhanced glycolysis and overexpress glucose transporter shows increased FDG uptake including malignant and activated inflammatory cells. Granulomatous disorder like cat scratch disease shows increased FDG uptake since granulomas histologically consist of mononuclear inflammatory cells or macrophages surrounded by lymphocytes. FDG PET/CT shows high diagnostic accuracy in primary ICC particularly the nodular or mass forming type [11]. Timely diagnosis of ICC is often challenging because of its silent clinical characteristics and low specificity of imaging modalities. Early-stage ICC may be asymptomatic, while at more advanced stages patients may experience weight loss, abdominal discomfort, malaise, jaundice, hepatomegaly, or a palpable mass [12,13]. Besides its notable metabolic activity, radiologic features of ICC comprise capsular retraction, peripheral biliary dilatation, satellite nodules, peripheral rim-like enhancement, central delayed enhancement, and irregular margins [11]. Even though tumor markers in this patient were within normal limits, tumor markers are often overlapping with other benign diseases and have low sensitivity for early-stage disease [13]. Furthermore, given that CSD cases are infrequently reported in Indonesia, we did not initially consider it as a potential diagnosis. Thus, with these findings pointing out to ICC, we decided to proceed with liver resection. For efficiency and according to NCCN 2023 about principle of gallbladder cancer, a pre-operative diagnostic biopsy is not necessary and a definitive resection should be carried out as long as resectable.
The underlying cause of CSD was revealed only after we received the biopsy result. When examining the specimen collected from the biopsy under a microscope using conventional haematoxylin and eosin staining, we observed indications of a chronic inflammatory granulomatous condition, which did not suggest a malignant condition. Upon analyzing the biopsy result, our initial suspicion leaned towards a mycobacterial infection, particularly Mycobacterium tuberculosis, which is prevalent in Indonesia. Consequently, we conducted a fast acid staining, which yielded negative results for mycobacterial infection. Additionally, microscopic examination of the specimen exhibited palisading histiocytes surrounding a suppurative necrotizing area, which more relevant to later phase of CSD [14].
Another test for B. henselae infection is serological testing. An immunofluorescence assay to detect the presence of IgM and IgG antibodies against B.henselae has higher sensitivity and specificity for confirming this diagnosis and identifying microorganisms on Warthin-Starry staining. Values higher than 1:64 for IgG and 1:15 for IgM are considered diagnostic for CSD [3]. Unfortunately, these tests are not routinely done in Indonesia and therefore are not performed in our patient.
Although the majority of CSD cases resolve spontaneously without the need for antimicrobial therapy, azithromycin has emerged as a common therapeutic choice for CSD, especially when dissemination to the liver, spleen, eye, or central nervous system occurs [1]. A study showed statistically significant improvement in patients treated with azithromycin [15]. Current recommendations for pediatric patients is a 5-day course of azithromycin (500 mg or 10 mg/kg/day as the initial dose, followed by 250 mg or 5 mg/kg/day for the remaining 4 days) [7]. We found that no recommendations for adult patients with CSD had been established. Moreover, the positve therapeutic outcome of using the preferred antibiotics in this patient, also further support the diagnostic of CSD.
In conclusions, CSD should be considered as a potential cause of liver abnormalities, especially in patients with a history of close contact with cats. Conventional histopathologic testing such as H&E staining could be useful for diagnosing CSD in medical facilities with limited resources, while also prospectively follow the progress of treatment using antibiotics. Although most cases of CSD resolve spontaneously, more severe cases can lead to serious complications and more aggressive treatment may be necessary.