저용량의 타목시펜 복용 후 발생한 급성 괴사성 췌장염
Hypertriglyceridemia-induced Severe Necrotizing Pancreatitis Following Low Dose Tamoxifen Administration
Article information
Abstract
51세 여자가 갑자기 발생한 심한 복통으로 응급실에 내원하였다. 환자는 1년 전 유방암으로 수술을 시행받았고 6개월 전부터 저용량(하루에 10 mg)의 타목시펜을 복용하기 시작했다. 환자의 복통은 중증의 궤사성 췌장염이 원인으로 밝혀졌다. 환자는 특별한 기저질환이나 약물 복용력은 없었으며 음주도 하지 않았다. 내시경 초음파에서 담석은 보이지 않았다. 환자의 6개월 전 중성지방의 수치는 정상이었으나 응급실 내원 당시의 수치는 2,534 mg/dL로 확인되었다. 고중성지방혈증과 췌장염의 원인으로 타목시펜이 가장 의심되었기에 타목시펜 중단 후 보존적 치료를 시행하였고 회복되어 퇴원하였다. 퇴원시 환자의 중성지방 수치는 정상이었다. 타목시펜은 유방암 치료에 효과적인 약이나 처방 후 지질대사에 대한 추적관찰과 관심이 반드시 필요하다.
Trans Abstract
A 51-year-old woman visited the emergency room with severe abdominal pain of acute onset. She had undergone a breast cancer operation one year previously and had been taking a half-dose (10 mg per day) of tamoxifen for 6 months. She was diagnosed with severe acute necrotizing pancreatitis. She had no other specific underlying disease or medical history. She did not drink alcohol and showed no gallstones on endoscopic ultrasound examination. Her blood triglyceride level had been normal before tamoxifen but had gradually increased to 2,534 mg/dL 6 months after beginning tamoxifen. Tamoxifen was regarded as a very likely causative factor for her necrotizing pancreatitis. After discontinuing the drug and receiving supportive care, she eventually recovered, and her blood triglyceride levels dropped to a normal range. Tamoxifen may be a useful drug for treating breast cancer, but doctors should pay attention to the patient’s blood triglyceride level during the medication regimen.
INTRODUCTION
Common causes of acute pancreatitis include gallstones, alcohol, hypertriglyceridemia, hypercalcemia, and drugs [1]. One cause of hypertriglyceridemia is the drug tamoxifen, an effective chemotherapeutic agent for various cancers that is most commonly used in the treatment of breast cancer. This drug is a selective estrogen receptor modulator but has a variety of metabolic effects, including elevation of triglycerides and chylomicrons. Tamoxifen-induced hypertriglyceridemia can lead to acute pancreatitis, and often reported [2-4]. But, most of these patients presented with mild acute pancreatitis and other causative factors, such as gallstones or alcohol consumption in addition to dyslipidemia. In this case, we report a patient who had no underlying risk factors for acute pancreatitis, but who presented with severe acute necrotizing pancreatitis after medication with low dose tamoxifen. Her pancreatitis was strongly thought to be related to hypertriglyceridemia caused by tamoxifen.
CASE
A 51-year-old post-menopausal female patient visited the Department of Emergency Medicine for severe epigastric pain. At the time of admission, her blood pressure was 92/66 mmHg, respiratory rate was 18 times per minute, heart rate was 88 beats per minute, and body temperature was 36.6℃. She was 161 cm tall and weighed 68 kg, with a body mass index of 26.2. She did not drink alcohol. She had a 15 pack-years smoking history. Eighteen years earlier, she had undergone breast-conserving surgery, followed by radiation therapy, for a right breast ductal carcinoma in situ. One year previously, a new palpable mass was found in the right remnant breast, and the recurrence of invasive carcinoma was confirmed. She underwent a total mastectomy and reconstruction. Her immunohistochemistry results showed estrogen receptor and progesterone receptor positivity and her C-erb-b2 was 2+ positive. Adjuvant chemotherapy with doxorubicin (60 mg/m2) and cyclophosphamide (600 mg/m2) was given every 3 weeks for 12 weeks after surgery. One month after chemotherapy, the patient started taking a half-dose of tamoxifen (10 mg per day). Her baseline blood triglyceride level was in the normal range of 132 mg/dL before tamoxifen administration, but she underwent a gradual elevation in blood triglyceride levels after beginning tamoxifen, reaching 292 mg/dL in 1 month and 558 mg/dL in 2 months, as noted in serial blood tests. By accident, no specific treatment for hypertriglyceridemia was given to this patient.
When the patient visited the emergency room, laboratory tests showed the following values: white blood cell count, 14,300 cells/μL with 79.3% neutrophils; hemoglobin, 12.9 g/dL; hematocrit, 36.3%; platelet count, 223,000 platelets/μL; aspartate aminotransferase: alanine aminotransferase level, 24:20 IU/L; total bilirubin level, 0.18 mg/dL; alkaline phosphatase level, 80 IU/L; protein:albumin level, 7.2:3.9 g/dL; prothrombin time (international normalized ratio), 10.2 seconds (0.90); serum amylase, 348 U/L; serum lipase, 945 U/L; serum lactate dehydrogenase, 263 U/L; and serum cholesterol, 428 mg/dL. Her serum sodium, potassium, chloride, blood urea nitrogen, creatinine, glucose, creatine kinase, and C-reactive protein were within normal limits. Computed tomography showed a diffuse swelling of the pancreatic parenchyma and extensive surrounding inflammatory fatty stranding, with an amount of necrotic fluid collection along the peri-pancreatic area, extending to the right mesocolon (Fig. 1). Laboratory tests performed on the day after admission showed a triglyceride level of 2,534 mg/dL, whereas her high-density-lipoprotein-cholesterol, low-density lipoprotein (LDL)-cholesterol, subclass immunoglobulin G4, anti-nuclear antibody, carcinoembryonic antigen, and hemoglobin A1C levels were in the normal range. She had no evidence of gallstones on endoscopic ultrasound examination (Fig. 2). The patient was diagnosed with hypertriglyceridemiainduced acute necrotizing pancreatitis. The cause of hypertriglyceridemia and acute pancreatitis was presumed to be tamoxifen. Tamoxifen was discontinued, and conservative treatment for acute pancreatitis was initiated. Fenofibrate (160 mg per day) was added to treat the patient’s hypertriglyceridemia. A follow-up computed tomography scan after 2 weeks showed extensive wall-off necrosis in the pancreatic head and mesocolon; however, the patient did not complain of any symptoms and was eventually discharged, with tamoxifen discontinued. Her triglyceride level at discharge was 186 mg/dL. The patients stopped fenofibrate after triglyceride normalization. After the restart of aromatase inhibitor letrozole (2.5 mg per day) as a risk of recurrence, triglyceride was elevated again and taking fenofibrate together as a treatment, and now maintains the normal level of triglyceride.
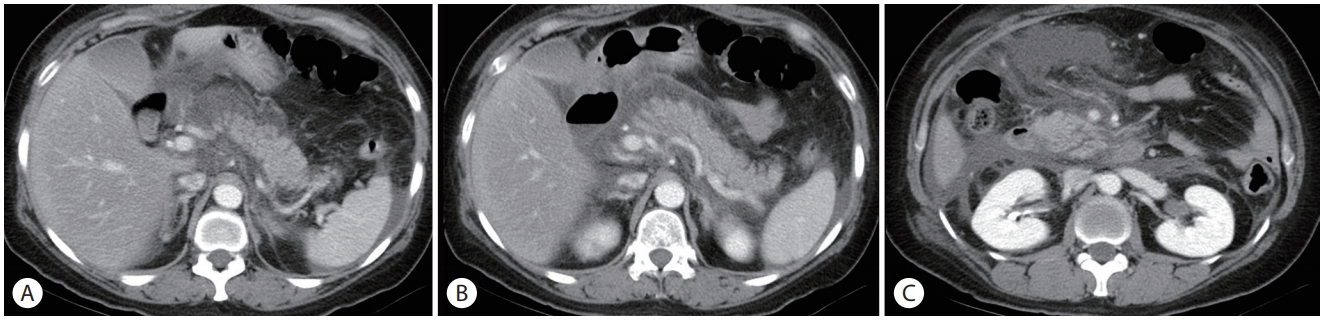
Computed tomography image of the pancreas. The pancreas shows diffuse swelling of the pancreatic parenchyma, as well as surrounding inflammatory fatty stranding with large amounts of fluid along the peri-pancreatic area. (A) Pancreas tail level. (B) Pancreas body level. (C) Pancreas head level.
DISCUSSION
Tamoxifen, a selective estrogen receptor modulator, is widely used for patients with breast cancer who are hormone-receptor positive because of its minimal side effects and good compliance [5]. Tamoxifen has some minor side effects, including nausea, vomiting, hot flashes, skin rashes, alopecia, and rarely thromboembolism. Tamoxifen acts as a hormone antagonist in the breast but has agonistic effect to estrogen in lipid metabolism, thus reducing LDL, total cholesterol, and lipoprotein(a). As a result, the synthesis of very low-density lipoprotein increases and triglyceride levels rise [6,7]. This change in lipid metabolism can cause acute pancreatitis and is often reported in the literature.
Acute pancreatitis is a reversible inflammatory disease of the pancreas and is a commonly encountered gastrointestinal disease in clinical practice. The causes of acute pancreatitis are primarily alcohol consumption and gallstones, but other metabolic diseases can contribute. Hypertriglyceridemia, especially with levels above 1,000 mg/dL, is well known as the cause of acute pancreatitis [1]. The mechanism by which triglyceride causes pancreatitis is not clear, but the accumulation of blood chylomicrons is thought to be involved. This accumulation causes hypertriglyceridemia, which raises the viscosity of the blood passing through the pancreas to produce a "sludge-like" effect. In addition, pancreatic lipases are thought to degrade triglycerides into free fatty acids that then cause cytotoxic injuries, resulting in an increase of inflammatory mediators that further exacerbates pancreatitis [8]. A literature review up to 2017 revealed 12 cases of acute pancreatitis emerging after tamoxifen administration. However, half of the reported cases had other risk factors for pancreatitis, such as dyslipidemia or gallstones, in addition to hypertriglyceridemia [2-4].
In our case, the patient had no past medical history of diabetes or gallstones. Her blood lipid status was normal before taking lose dose tamoxifen but gradually elevated after taking the medication. None of the other drugs the patient was taking were directly related to acute pancreatitis. Therefore, we considered tamoxifen to be the agent that induced hypertriglyceridemia and led to acute pancreatitis in this patient.
If hypertriglyceridemia or acute pancreatitis develops after taking tamoxifen, some clinicians recommend that the dose be reduced to 10 mg per day [9]. In fact, hypertriglyceridemia occurred at a dose of 20 mg per day in most of the reported cases [2-4]. The existing literature reports only one patient who took 10 mg tamoxifen per day and presented with acute pancreatitis; however, that patient’s pancreatitis was very mild at the time of diagnosis [10]. In the present case, however, a life-threatening and necrotizing pancreatitis developed at only a daily dose of 10 mg, despite the absence of any specific underlying disease or risk factor, which suggests that drug dose does not correlate with pancreatitis severity. We suggest careful monitoring of the blood triglyceride levels in patients prescribed tamoxifen, regardless of drug dosage.
Previous studies have reported that patients without hyperlipidemia at the baseline can be prescribed letrozole, an aromatase inhibitor, since it has less appreciable adverse effects on serum lipid levels [11]. Therefore, if patients with hypertriglyceridemia or acute pancreatitis after tamoxifen use are in high-risk groups for breast cancer recurrence, letrozole may be considered. However, this drug may also cause dyslipidemia, hypoestrogenism and osteoporosis are also important side effects [11,12]. So, Tamoxifen may be recommended for hormone therapy in breast cancer patients who are positive for hormone receptors in premenopausal women [12]. Overall, our results suggest that the most important considerations are performing periodic examinations and remaining aware of the possible elevation of lipid levels before and after taking tamoxifen, regardless of other clinical risk factors.
In conclusion, tamoxifen is well known as a useful drug for the treatment of various cancers. Clinicians should keep in mind that tamoxifen can cause hypertriglyceridemia, which can be associated with life-threatening pancreatitis. Even if the patient is taking a low dose of tamoxifen or has no other risk factors for pancreatitis, periodic lipid blood tests must be performed before and after tamoxifen therapy. If abnormal findings are found, medical interventions (e.g., statin or fenofibrate) and lifestyle modifications (e.g., diet therapy) should be considered.
Notes
Conflict of Interest
The authors have no conflicts to disclose.

